Accelerating Development of TB Treatment
The Preclinical Design and Clinical Translation of Regimens for Tuberculosis (PReDiCTR-TB) Consortium unites top scientists and global stakeholders to prioritize and advance the most promising TB regimens into clinical trials. Our data-driven translational research platform bridges preclinical models with clinical success to identify TB regimens with the greatest potential for clinical success.

How We Work
The Challenge
Treatment for TB requires multidrug therapy, significantly complicating regimen design due to the sheer number of possible drug combinations (on the order of tens of thousands of potential combinations).
Traditional development is slow, costly, and often fails to meet the needs of vulnerable populations. It has often been hindered and misled by poor preclinical-to-clinical translation. With so many possible combinations, the challenge becomes prioritizing the most promising multidrug regimens to be tested in resource-intensive clinical trials.


– Rada Savic, PhD, MBA
Our Approach
The PReDiCTR-TB Consortium unites key scientists and dedicated stakeholders to build a translational research platform to accurately and efficiently identify and advance the most promising TB treatment regimens through the developmental pipeline into clinical trials.
- Establish a multidisciplinary coalition of scientific leaders to develop and pursue a translational research agenda to evaluate, prioritize and optimize novel drug regimens for clinical trials.
- Validate, refine and utilize a model-based platform for quantitative predictions of clinical outcomes to evaluate, prioritize, and optimize.
- Validate, refine, and utilize in vitro methodologies to predict in vivo efficacy of regimens.


PReDiCTR-TB Consortium Researchers
.avif)
It's not just moving these regimens forwards into trials but backtranslating them into better preclinical research.
Innovation
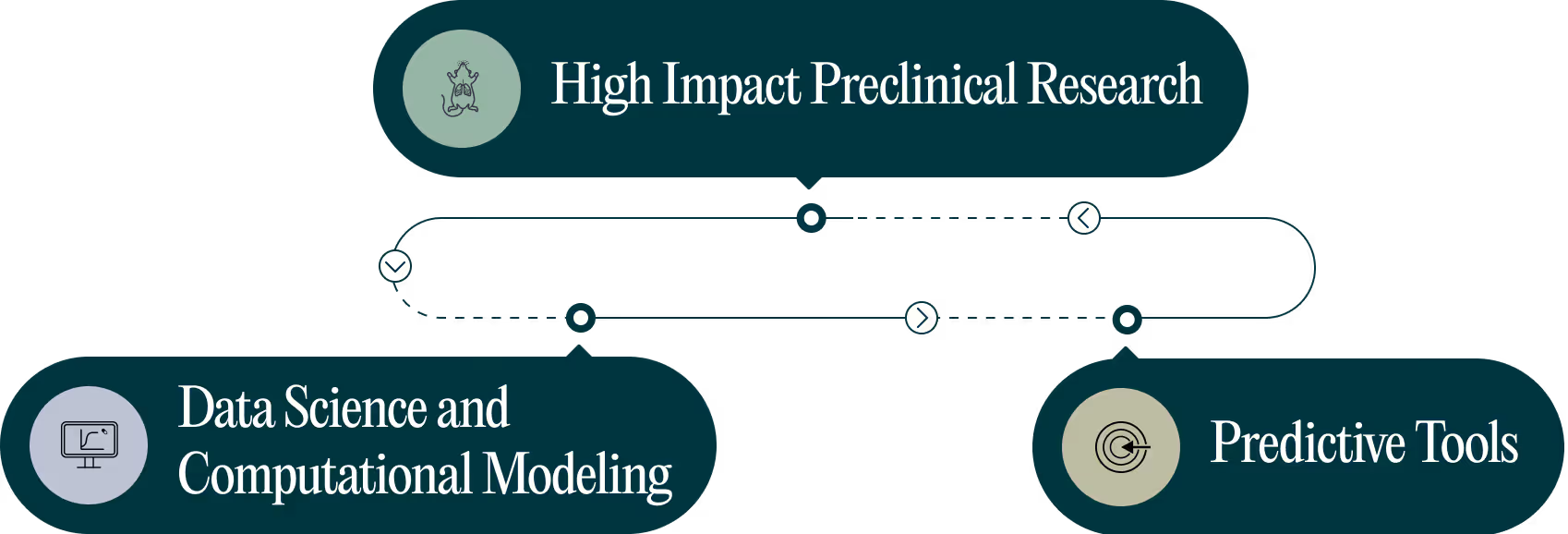
We now have the knowledge and tools to apply to the design of novel regimens to streamline TB regimen development, for all. We have all the necessary parts to integrate the experiments, knowledge and data, to carefully design and select the best regimens to advance further.
Integrated approach for backtranslation of existing regimens. Forward prediction and regimen ranking of novel regimens.
- Better represents high-risk patient phenotypes
- Enables lesion pharmacokinetic-pharmacodynamic (PK-PD) assessment
- Efficient ranking of regimens to test in the relapsing mouse model
- May be applicable to C3HeB/FeJ mice
- Efficiently prioritize regimens for in vivo study
- Can include PK-informed dose ratios
- Can include lesion PK-informed dose ratios
- Can be studied in key milieu




30+
20+
The Consortium
The PReDiCTR-TB Consortium is a collaboration of 30+ researchers, 20+ institutions, and multiple clinical trial networks working across the TB drug development pipeline.

Collaborating with Global Networks
Explore Our Preclinical & Data Sciences Laboratories
.avif)

Omics
Validate, refine & utilize in vitro methodologies to predict in vivo efficacy of regimens
.avif)
.svg)
PhyBM
Physiologic Biomarker, synthesizing in vitro, murine & human evidence

.svg)
DSMG
Data integration and clinical simulation tools to advance drug therapy
.avif)
.svg)
DMPK
Incorporating lesion-centric PK and PD measurements into translational models

.svg)
CSU-mouse
Optimizing translational value of preclinical mouse models
.avif)
.svg)
DiaMOND
Systematic in vitro studies of drug combinations
.avif)

JHU-mouse
Murine models to inform TB regimen development
Explore the Environments Where Innovation Begins
The Core PReDiCTR-TB Consortium Researchers
Introducing The Consortium Scientists

Rada Savic
,
PhD, MBA

Research in the Savic Lab focuses on data integration and the transformative potential of computational tools and artificial intelligence to advance drug development and routine drug therapy in infectious diseases including special populations such as children and pregnant women. Dr. Savic applies innovative, quantitative, and systems pharmacology methods to propose optimized and precision dosing strategies, data-driven risk stratification algorithms, and inspires new clinical simulation tools across the infectious disease landscape.
%20(1).avif)
Eric Nuermberger
,
MD
.svg)
Research in the Nuermberger lab focuses primarily on experimental chemotherapy for tuberculosis. We use proven murine models of active and latent tuberculosis infection to assess the effectiveness of novel antimicrobials. A key goal is to identify new agents to combine with existing drugs to shorten tuberculosis therapy or enable less frequent drug administration. We're also using a flow-controlled in vitro pharmacodynamic system to better understand the pharmacodynamics of drug efficacy and the selection of drug-resistant mutants during exposure to current agents.
%20(1).avif)
Gregory Robertson
,
PhD
.svg)
Dr. Robertson’s research is focused on the field of Mycobacterium tuberculosis host-pathogen interactions with emphasis on the development of novel preclinical tools, use of TB mouse models for drug evaluation, and evaluation of drug activity and resistance in vitro and in vivo. He also leads preclinical animal studies as part of the Consortium for Applied Microbial Metrics including other investigators from the University of California San Francisco, Denver VA Medical Center, and CU Anschutz Medical Campus.
As part of the PReDiCTR-TB Consortium, the CSU-Mouse Preclinical Laboratory (CSU-Mouse PL) focuses on optimizing translational value of preclinical mouse models.
%20(1).avif)
Nicholas Walter
,
MD, PhD
.svg)
Dr Walter’s research focuses on development and practical application of novel pharmacodynamic markers with the goal of accelerating new regimen development. Dr Walter’s team has developed a portfolio of molecular tools that provide information on the pathogen’s physiologic processes (i.e., “pathogen health”) rather than pathogen burden.
This orthogonal perspective reveals insights distinct from traditional culture-based pharmacodynamics. The Walter lab applies these tools in conjunction with other PReDiCTR-TB labs to maximize information available for decision-making.
.avif)
Jansy Sarathy
,
PhD

Jansy Sarathy is an Assistant Professor at the Hackensack Meridian School of Medicine in Nutley, New Jersey, and an Assistant Member at the Center for Discovery and Innovation. Her research focuses on applying site-of-disease pharmacokinetic-pharmacodynamic (PK-PD) concepts to rationalize TB treatment efficacy. She is particularly interested in addressing nonreplicating persistent populations of M. tuberculosis in hard-to-treat sites of infection.
%20(1).avif)
Martin Voskuil
,
PhD
.svg)
Dr. Voskuil’s research is focused on the ability of aerobic bacterial pathogens to exist in non-replicating states that are distinct from the replicating bacteria most often studied. We hypothesize that these states are central to the pathogens survival during latent infections and their ability to tolerate conventional drug treatment. The physiological state of Mycobacterium tuberculosis during latent infection has been a focus of our research. A primary reason for the continued threat from M. tuberculosis lies in its ability to establish an asymptomatic latent infection, which serves as a reservoir for future infections.
.avif)
Dirk Schnappinger
,
PhD
.svg)
The Schnappinger Lab studies Mycobacterium tuberculosis (Mtb) with the goal to facilitate the development of new medicines for treatment or prevention of Tuberculosis (TB). This work began with developing a regulatory system that allows to turn Mtb genes on and off, both in vitro and during infections. The Schnappinger lab now uses this system (i) to evaluate Mtb gene products as new targets for TB drug development by documenting the impact of their inactivation on growth and persistence of Mtb in vitro and during infections; (ii) to help elucidate the mechanisms by which small-molecules inhibit the growth of Mtb.
%20(1).avif)
Jeremy Rock
,
PhD
.svg)
Mycobacterium tuberculosis is the leading cause of death due to infectious disease. By investigating the mechanisms that enable this bacterium to cause tuberculosis and evade current antibiotics, the Rock lab, at The Rockefeller University, aims to lay the foundation for new therapeutic strategies to improve control of this epidemic.
Despite the discovery of antibiotics, tuberculosis (TB) remains an enduring global public health threat.
%20(1).avif)
Kelly Dooley
,
MD, PhD, MPH
.avif)
Trained as an Infectious Diseases specialist and Clinical Pharmacologist, Dr. Dooley’s research focuses on tuberculosis therapeutics with an emphasis on clinical trials of TB and HIV-TB co-treatment. She has served as lead investigator for trials of therapeutics for drug-sensitive and drug-resistant TB, HIV-TB, and pediatric TB meningitis funded by the NIH, the FDA, UNITAID, and industry partners.
.avif)
Bree Aldridge
,
PhD

Our research focuses on designing optimized therapies for TB using cell biology and engineering approaches. Our lab is a multidisciplinary research team, integrating quantitative measurement with computational modeling and analysis to create intuitive descriptions of complex cell biology. We focus our studies on (1) characterizing single-cell determinants of mycobacterial drug tolerance, (2) understanding how growth heterogeneity is controlled, and (3) engineering combination therapy.
As part of the PReDiCTR-TB Consortium, the DiaMOND Preclinical Laboratory (DiaMOND PL) uses a geometric optimization of traditional drug combination assays, to efficiently measure drug interactions and combination efficacies.
.avif)
Sabine Ehrt
,
PhD
.svg)
Mycobacterium tuberculosis is responsible for the largest number of deaths worldwide due to a single bacterial infection. The increasing number of multi-drug resistant (MDR) and extensively drug resistant (XDR) M. tuberculosis strains and the lack of an effective vaccine exacerbate the problem. The research in our laboratory focuses on the interaction of M. tuberculosis with macrophages, aims to elucidate the bacterial and host molecular mechanisms required to establish and maintain latent tuberculosis infection and seeks to facilitate the development of new therapies for the treatment and prevention of tuberculosis.
%20(1).avif)
Kyu Rhee
,
MD, PhD
.svg)
Dr. Rhee currently serves as professor of medicine in the Division of Infectious Diseases, associate professor of microbiology and immunology at Weill Cornell and associate attending physician at NewYork-Presbyterian Hospital/Weill Cornell Medical Center. His clinical interests are in the areas of tuberculosis and antibiotic pharmacology. His major research interests are in elucidating the basic physiology of Mycobacterium tuberculosis, the causative agent of TB, and identification of novel drug targets.
Our Latest Publications
Early Career Seminar Series: Designing Better TB Regimens
Stay tuned for announcements about upcoming seminars in this series.

Let’s Transform
TB Treatment Together
Interested in collaborating or learning more about our work?




%20(1).avif)

.avif)
.avif)

%20(1).avif)

%20(1).avif)
%20(1).avif)

%20(1).avif)

%20(1).avif)



%20(1).avif)

%20(1).avif)
.avif)